Metabolism of 2024 FDA approved small molecules part 1
By Julia Shanu-Wilson
Involvement of active metabolites
The number of small molecules approved by the FDA in 2024 totaled 31 out of 50 NMEs with 56% (28) of these comprising small molecule therapies. Of these, 27 were analysed for their routes of metabolism.
It is noticeable that there is less reliance on only Phase 1 routes of metabolism in this set of drugs, compared to the past two years (Figure 1). Several biotransformation pathways were reported as relevant, including oxidation by cytochrome P450s, aldehyde oxidase and flavin-containing monooxygenases; hydrolysis; cytosolic reduction; glucuronidation; glycosylation; glutathione conjugation; sulfation; and more atypical routes. Of those drugs metabolized by CYPs, enzymes involved included CYP3A4 (55%) as well as CYP1A2, CYP2C8, CYP2C9, and CYP2D6. As a proportion of overall metabolic routes, CYP3A4 as a major route was noticeably lower, with Phase II and hydrolytic routes featuring more than in previous years. A table summarising all the cited routes of biotransformation will be provided in a future instalment.
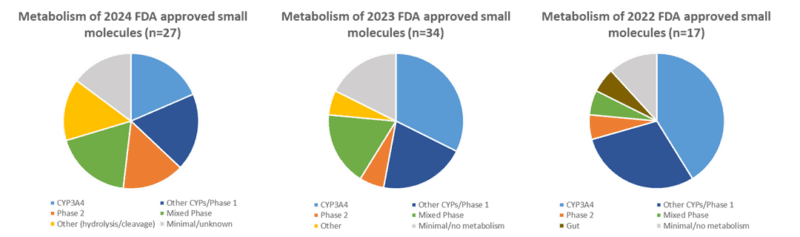
Figure 1: Comparison of the major routes of metabolism of small molecule drugs approved by the FDA in 2024, 2023 and 2022
A superior drug based on metabolism of an existing drug
Aprocitentan (Tryvio; ACT-132577) is an endothelin (ET) A and B receptor antagonist approved for treatment of hypertension, where high blood pressure cannot be adequately controlled by other drugs. It is metabolized primarily by UGT1A1- and UGT2B7-mediated N-glucosidation and hydrolysis (Figure 2), although five different metabolic pathways are involved [1]. These comprise hydrolysis to M1, glucosidation to M3, glucuronidation to M8, oxidative debromination then glucuronidation to M10, and N-oxidation at the pyrimidine sulfamide moiety to M9. M9 and other metabolites subsequently undergo glucuronidation. All human metabolites were detected in the rat and/or the dog and no disproportionate human metabolites were identified.
In an in vitro setting, formation of M1 results from both chemical and enzymatic hydrolysis. M1 can then be metabolized to secondary metabolites M2 and M5 through CYP/FMO-dependent reactions. M2 undergoes further glucuronidation to M4, and M5 is oxidized to M6 in human hepatocytes.
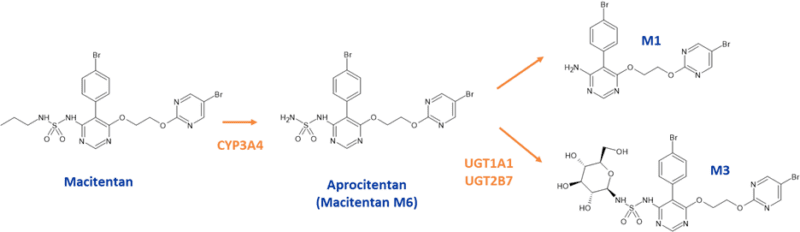
Figure 2: Main metabolic routes of macitentan and aprocitentan
Whilst aprocitentan is not metabolized to an active metabolite, it itself is the active metabolite M6 of the drug macitentan (Opsumit). M6 is generated through oxidative depropylation of the sulfamide group of macitentan, mainly by CYP3A4 [2]. Although aprocitentan has a lower affinity for the ET receptors than its parent compound, it maintains higher plasma concentrations than macitentan [2]. Another advantage afforded by aprocitentan’s independence of CYP-mediated metabolism, is that the drug has limited potential for interactions with drugs that induce or inhibit CYPs [3]. As such, aprocitentan can be administered simultaneously with any CYP inhibitor or inducer, with no need for dose adjustment [4].
QTc involvement of an active metabolite
Revumenib (Revuforji) belongs to a new class of drugs called menin inhibitors and was approved for treatment of KMT2A-rearranged leukemias. CYP3A4 is involved in generation of an active metabolite (M1) of the drug, although the structure of M1 and other secondary metabolites have not been reported in the literature.
 Interestingly, although M1 does not contribute to its efficacy at the approved recommended dosage, it does contribute to revumenib’s clinically significant effects on QTc [5]. As such, strong or moderate CYP3A4 inducers may increase metabolite (M1) exposure and risk of QTc interval prolongation. During clinical trials, QTc prolongation emerged as the primary dose-limiting adverse event, but was reported to be manageable [6].
Interestingly, although M1 does not contribute to its efficacy at the approved recommended dosage, it does contribute to revumenib’s clinically significant effects on QTc [5]. As such, strong or moderate CYP3A4 inducers may increase metabolite (M1) exposure and risk of QTc interval prolongation. During clinical trials, QTc prolongation emerged as the primary dose-limiting adverse event, but was reported to be manageable [6].
A major active metabolite formed by an atypical biotransformation pathway
The story behind the formation of Elafibranor’s major equipotent active metabolite is rather interesting as it involves an enzyme not normally implicated in drug metabolism. Elafibranor (Iqirvo) is a dual PPARα/δ agonist approved for treatment of primary biliary cholangitis. It is extensively metabolized to form the active metabolite, GFT1007 (M3), the mean systemic exposure (AUC) of which was 2 to 4-fold higher than that of the parent drug at steady state [7]. A major (inactive) acyl glucuronide GFT3351 also circulated in plasma (Figure 3).
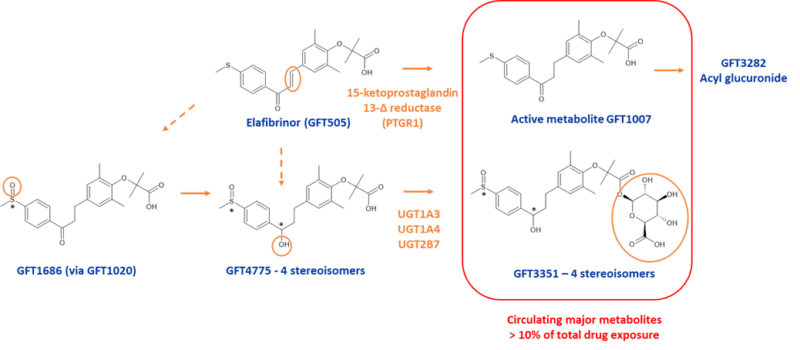
Figure 3: Proposed in vitro metabolic pathway for elafibranor adapted from Figure 25 in [7]; human and monkey hepatocyte incubations produce the widest range of metabolites
In vitro studies showed that elafibranor (GFT505) was metabolized to GFT1007 by a cytosolic enzyme, 15-ketoprostaglandin 13-Δ reductase (PTGR1). The active metabolite GFT1007 was further metabolized by CYP2C8, and UGT1A3 and UGT2B7.
Other enzymes involved in elafibranor’s metabolism include CYP2J2 and multiple UGTs (UGT1A3, UGT1A4, and UGT2B7). Formation of four stereoisomers of the acyl glucuronide (GFT3351) are observed, with two of these (GFT4825 and GFT4826) exceeding 10% of total drug exposure.
A major active CYP2C8-mediated metabolite
The final drug featured in this instalment is resmetirom (Rezdiffra), a thyroid hormone receptor beta (NR1A2) agonist, and the first drug approved to treat NASH (non-alcoholic steatohepatitis).
 Resmetirom is metabolized solely by CYP2C8 to a major disproportionate active metabolite in plasma, MGL-3623 (M1). Although MGL-3623 has a 28-times lower potency for THR-β than the parent drug, it represents 33% to 51% of resmetirom AUC at steady state following administration of 100 mg once daily [8]. The structure of MGL-3623 was included in the patent EP 4 112 056 A1 [9].
Resmetirom is metabolized solely by CYP2C8 to a major disproportionate active metabolite in plasma, MGL-3623 (M1). Although MGL-3623 has a 28-times lower potency for THR-β than the parent drug, it represents 33% to 51% of resmetirom AUC at steady state following administration of 100 mg once daily [8]. The structure of MGL-3623 was included in the patent EP 4 112 056 A1 [9].
Due to its prominence, the nonclinical safety profile of MGL-3623 was evaluated in a 3-month mouse toxicology study, genetic toxicology studies (Ames test, in vitro micronucleus assay in TK6 human lymphoblast cells, and in vivo rat micronucleus assay), an embryo-fetal development (EFD) study in rats, and a 6-month carcinogenicity study in transgenic mice, alongside the parent drug.
MGL3623 was not mutagenic in the Ames test or in the in vivo rat micronucleus assay, but was positive for induction of micronuclei in TK6 human lymphoblast cells with metabolic activation. However, based on comments in ICH guidance S2(R1) (Note 9) for in vitro cytogenetic assays, the relevance of the positive finding in the current study is uncertain because the increase in micronuclei was limited to a single concentration with cytotoxicity exceeding 50% (i.e., 59% growth inhibition was observed). MGL-3623 was therefore considered not to pose a safety concern.
Due to metabolism by CYP2C8 and not by any other CYPs, concomitant use of resmetirom is not recommended with strong CYP2C8 inhibitors such as gemfibrozil, and dosage reduced with moderate inhibitors, e.g., clopidogrel.
What’s next?
Stay tuned for our next instalment in which we take a look at 2024 FDA-approved drugs incorporating deuterium in order to manage metabolism, as well as those drugs primarily metabolized through glucuronidation.
If you have a metabolite you would like to access or identify, please contact us us at Hypha to discuss.
Acknowledgements
Many thanks to Chris de Savi for compiling the list of FDA Novel Drug Approvals 2024 on LinkedIn.
Other useful reading at:
https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2024
https://drughunter.com/articles/2024-novel-small-molecule-fda-drug-approvals
https://cen.acs.org/pharmaceuticals/50-new-drugs-received-FDA/103/i2
References
[1] Center for Drug Evaluation and Research Application Number: 217686Orig1s000 Integrated Review. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2024/217686Orig1s000IntegratedR.pdf
[2] Sidharta PN, Treiber A, Dingemanse J. Clinical pharmacokinetics and pharmacodynamics of the endothelin receptor antagonist macitentan. Clin Pharmacokinet. 2015 May;54(5):457-71. doi: 10.1007/s40262-015-0255-5.
[3] Angeli F, Verdecchia P, Reboldi G. Aprocitentan. A Dual Endothelin Receptor Antagonist Under Development for the Treatment of Resistant Hypertension. Cardiol Ther 10, 397–406 (2021). https://doi.org/10.1007/s40119-021-00233-7
[4] Yao Y, Fan B, Yang B, Jia Z, Li B. Aprocitentan: A new development of resistant hypertension. J Clin Hypertens. 2023; 25: 587–590. https://doi.org/10.1111/jch.14686
[5] Highlights of prescribing information. Revuforji. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/218944s000lbl.pdf
[6] Salman MY, Stein EM. Revumenib for patients with acute leukemia: a new tool for differentiation therapy. Haematologica. 2024 Nov 1;109(11):3488-3495. doi: 10.3324/haematol.2022.282621.
[7] Center for Drug Evaluation and Research Application Number: 218860Orig1s000 Integrated Review. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2024/218860Orig1s000IntegratedR.pdf
[8] Center for Drug Evaluation and Research Application Number: 217785Orig1s000 Integrated Review https://www.accessdata.fda.gov/drugsatfda_docs/nda/2024/217785Orig1s000IntegratedR.pdf
[9] Methods of treating liver disorders or lipid disorders with a THR-Beta agonist. Patent EP 4 112 056 A1. https://data.epo.org/publication-server/rest/v1.2/publication-dates/20230104/patents/EP4112056NWA1/document.pdf